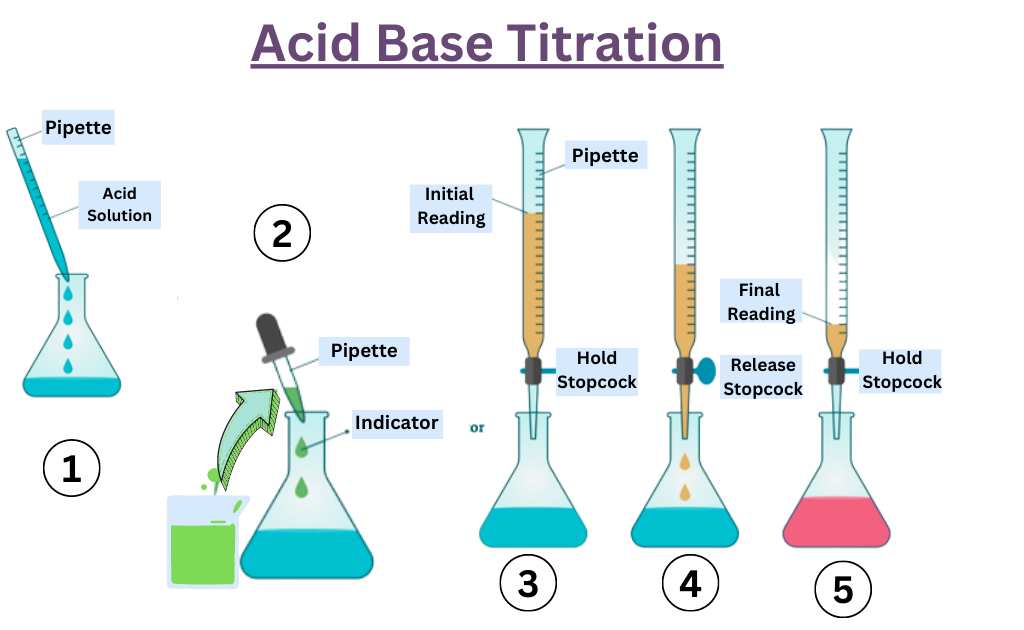
Indicator Work In A Titration . Indicators are substances whose solutions change color due to changes in ph. The graph shows the results obtained using two indicators (methyl red and. In the case of phenolphthalein, the protonated form is colorless, while the. The ph range of phenolphthalein is about 8.3 to 10.0, but the. This page assumes that you know. To be useful as an indicator, the two forms must be different colors. They are usually weak acids or bases,. The two most common indicators that are used in titrations are methyl orange and phenolphthalein.
from themasterchemistry.com
This page assumes that you know. The ph range of phenolphthalein is about 8.3 to 10.0, but the. Indicators are substances whose solutions change color due to changes in ph. The graph shows the results obtained using two indicators (methyl red and. To be useful as an indicator, the two forms must be different colors. In the case of phenolphthalein, the protonated form is colorless, while the. They are usually weak acids or bases,. The two most common indicators that are used in titrations are methyl orange and phenolphthalein.
Acid Base TitrationWorking Principle, Process, Types And Indicators
Indicator Work In A Titration The ph range of phenolphthalein is about 8.3 to 10.0, but the. To be useful as an indicator, the two forms must be different colors. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. They are usually weak acids or bases,. Indicators are substances whose solutions change color due to changes in ph. In the case of phenolphthalein, the protonated form is colorless, while the. This page assumes that you know. The graph shows the results obtained using two indicators (methyl red and. The ph range of phenolphthalein is about 8.3 to 10.0, but the.
From www.compoundchem.com
Chemistry Techniques Titration Compound Interest Indicator Work In A Titration The two most common indicators that are used in titrations are methyl orange and phenolphthalein. Indicators are substances whose solutions change color due to changes in ph. They are usually weak acids or bases,. The graph shows the results obtained using two indicators (methyl red and. In the case of phenolphthalein, the protonated form is colorless, while the. To be. Indicator Work In A Titration.
From franco-krussell.blogspot.com
How to Determine Which Indicator to Use for Titration Indicator Work In A Titration In the case of phenolphthalein, the protonated form is colorless, while the. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. Indicators are substances whose solutions change color due to changes in ph. They are usually weak acids or bases,. The graph shows the results obtained using two indicators (methyl red and. The ph. Indicator Work In A Titration.
From www.reagent.co.uk
How is Titration Used in the Pharmaceutical Industry? Indicator Work In A Titration This page assumes that you know. The graph shows the results obtained using two indicators (methyl red and. In the case of phenolphthalein, the protonated form is colorless, while the. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. Indicators are substances whose solutions change color due to changes in ph. The ph range. Indicator Work In A Titration.
From www.slideserve.com
PPT Titrations PowerPoint Presentation, free download ID5572517 Indicator Work In A Titration In the case of phenolphthalein, the protonated form is colorless, while the. This page assumes that you know. Indicators are substances whose solutions change color due to changes in ph. To be useful as an indicator, the two forms must be different colors. The graph shows the results obtained using two indicators (methyl red and. The ph range of phenolphthalein. Indicator Work In A Titration.
From www.youtube.com
Titration curves and acidbase indicators Chemistry Khan Academy Indicator Work In A Titration They are usually weak acids or bases,. This page assumes that you know. To be useful as an indicator, the two forms must be different colors. Indicators are substances whose solutions change color due to changes in ph. The ph range of phenolphthalein is about 8.3 to 10.0, but the. The two most common indicators that are used in titrations. Indicator Work In A Titration.
From slideplayer.com
Titration Introduction Experiment ppt download Indicator Work In A Titration This page assumes that you know. To be useful as an indicator, the two forms must be different colors. The graph shows the results obtained using two indicators (methyl red and. Indicators are substances whose solutions change color due to changes in ph. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. The ph. Indicator Work In A Titration.
From luz-has-richmond.blogspot.com
How Tknow Which Indicator to Use for Titrations LuzhasRichmond Indicator Work In A Titration The ph range of phenolphthalein is about 8.3 to 10.0, but the. In the case of phenolphthalein, the protonated form is colorless, while the. This page assumes that you know. To be useful as an indicator, the two forms must be different colors. The graph shows the results obtained using two indicators (methyl red and. They are usually weak acids. Indicator Work In A Titration.
From www.studocu.com
Experiment 7 Titrations Experiment 7 Titrations Required reading Indicator Work In A Titration The two most common indicators that are used in titrations are methyl orange and phenolphthalein. The ph range of phenolphthalein is about 8.3 to 10.0, but the. Indicators are substances whose solutions change color due to changes in ph. The graph shows the results obtained using two indicators (methyl red and. This page assumes that you know. To be useful. Indicator Work In A Titration.
From www.vrogue.co
Ph Indicators Titration Curves Teaching Resources vrogue.co Indicator Work In A Titration The graph shows the results obtained using two indicators (methyl red and. The ph range of phenolphthalein is about 8.3 to 10.0, but the. Indicators are substances whose solutions change color due to changes in ph. They are usually weak acids or bases,. To be useful as an indicator, the two forms must be different colors. In the case of. Indicator Work In A Titration.
From themasterchemistry.com
Acid Base TitrationWorking Principle, Process, Types And Indicators Indicator Work In A Titration In the case of phenolphthalein, the protonated form is colorless, while the. The graph shows the results obtained using two indicators (methyl red and. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. They are usually weak acids or bases,. This page assumes that you know. Indicators are substances whose solutions change color due. Indicator Work In A Titration.
From facts.net
8 Captivating Facts About AcidBase Titration Indicator Work In A Titration They are usually weak acids or bases,. In the case of phenolphthalein, the protonated form is colorless, while the. The graph shows the results obtained using two indicators (methyl red and. The ph range of phenolphthalein is about 8.3 to 10.0, but the. Indicators are substances whose solutions change color due to changes in ph. To be useful as an. Indicator Work In A Titration.
From chem.libretexts.org
9.1 Overview of Titrimetry Chemistry LibreTexts Indicator Work In A Titration The ph range of phenolphthalein is about 8.3 to 10.0, but the. Indicators are substances whose solutions change color due to changes in ph. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. This page assumes that you know. In the case of phenolphthalein, the protonated form is colorless, while the. They are usually. Indicator Work In A Titration.
From chem.libretexts.org
AcidBase Titrations Chemistry LibreTexts Indicator Work In A Titration The graph shows the results obtained using two indicators (methyl red and. In the case of phenolphthalein, the protonated form is colorless, while the. This page assumes that you know. To be useful as an indicator, the two forms must be different colors. They are usually weak acids or bases,. The ph range of phenolphthalein is about 8.3 to 10.0,. Indicator Work In A Titration.
From vanesfirstworldproblems.blogspot.com
Vane's First World Problems Titrations Indicator Work In A Titration The graph shows the results obtained using two indicators (methyl red and. They are usually weak acids or bases,. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. This page assumes that you know. In the case of phenolphthalein, the protonated form is colorless, while the. To be useful as an indicator, the two. Indicator Work In A Titration.
From fity.club
Rhodanine Indicator Cyanide Titration Indicator Work In A Titration The two most common indicators that are used in titrations are methyl orange and phenolphthalein. The ph range of phenolphthalein is about 8.3 to 10.0, but the. This page assumes that you know. They are usually weak acids or bases,. The graph shows the results obtained using two indicators (methyl red and. To be useful as an indicator, the two. Indicator Work In A Titration.
From courses.lumenlearning.com
AcidBase Titrations Chemistry for Majors Indicator Work In A Titration In the case of phenolphthalein, the protonated form is colorless, while the. To be useful as an indicator, the two forms must be different colors. The graph shows the results obtained using two indicators (methyl red and. This page assumes that you know. They are usually weak acids or bases,. The two most common indicators that are used in titrations. Indicator Work In A Titration.
From www.scribd.com
Topic 12 Exercise 4 Titrations and Indicators Indicator Work In A Titration They are usually weak acids or bases,. The graph shows the results obtained using two indicators (methyl red and. Indicators are substances whose solutions change color due to changes in ph. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. The ph range of phenolphthalein is about 8.3 to 10.0, but the. This page. Indicator Work In A Titration.
From www.chemicals.co.uk
Titration Experiments In Chemistry The Chemistry Blog Indicator Work In A Titration Indicators are substances whose solutions change color due to changes in ph. The two most common indicators that are used in titrations are methyl orange and phenolphthalein. To be useful as an indicator, the two forms must be different colors. They are usually weak acids or bases,. In the case of phenolphthalein, the protonated form is colorless, while the. The. Indicator Work In A Titration.